Lec 7 - Quantum Mechanical Kinetic Energy
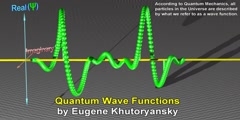
"Lec 7 - Quantum Mechanical Kinetic Energy" Freshman Organic Chemistry (CHEM 125) After pointing out several discrepancies between electron difference density results and Lewis bonding theory, the course proceeds to quantum mechanics in search of a fundamental understanding of chemical bonding. The wave function ψ, which beginning students find confusing, was equally confusing to the physicists who created quantum mechanics. The Schrödinger equation reckons kinetic energy through the shape of ψ. When ψ curves toward zero, kinetic energy is positive; but when it curves away, kinetic energy is negative! 00:00 - Chapter 1. Limits of the Lewis Bonding Theory 08:35 - Chapter 2. Introduction to Quantum Mechanics 16:36 - Chapter 3. Understanding Psi as a Function of Position 33:24 - Chapter 4. Understanding Negative Kinetic Energy and Finding Potential Energy Complete course materials are available at the Open Yale Courses website: http://open.yale.edu/courses This course was recorded in Fall 2008.
Video is embedded from external source so embedding is not available.
Video is embedded from external source so download is not available.
Channels: Chemistry (General)
Tags: Curvature Kinetic Energy Quantum Mechanics Schrödinger Wave Equation Wave function
Uploaded by: yalefreshorganic ( Send Message ) on 05-09-2012.
Duration: 49m 18s
Here is the next lecture for this course
Lec 8 - One-Dimensional Wave Functions
49:51 | 3836 viewsLec 21- Even further Orbital Angular Mome ...
52:10 | 3858 viewsQuantum wave functions
11:23 | 9667 viewsChemical Science - Matter As a Wave - Lec ...
44:24 | 20951 viewsLec 72 - Molar Kinetic Energy (Quiz)
01:45 | 3083 viewsLec 33 - Conformational Energy and Molecu ...
47:53 | 4179 viewsAtom wave functions
02:32 | 8266 viewsChemical Science - Free Energy of Formati ...
47:47 | 20420 viewsChemical Science-Atomic Theory of Matter ...
41:59 | 31802 viewsChemical Science - Particle-like Nature o ...
47:32 | 20670 viewsQuantum Mechanics: Properties Of Elementa ...
06:14 | 13334 viewsQuantum Mechanics Feynman Diagram
06:30 | 11077 viewsLec 1- Introduction to Quantum Mechanics, ...
44:05 | 9706 viewsLec 1 - Modern Physics: Quantum Mechanics ...
01:51:11 | 5399 viewsLec 2 - Modern Physics: Quantum Mechanics ...
01:51:04 | 4362 viewsNo content is added to this lecture.
This video is a part of a lecture series from of Yale
Lecture list for this course
Lec 2 - Force Laws, Lewis Structures and Resonance
Lec 3 - Double Minima, Earnshaw's Theorem and Plum-Puddings
Lec 4 - Coping with Smallness and Scanning Probe Microscopy
Lec 6 - Seeing Bonds by Electron Difference Density
Lec 8 - One-Dimensional Wave Functions
Lec 9 - Chladni Figures and One-Electron Atoms
Lec 10 - Reality and the Orbital Approximation
Lec 11 - Orbital Correction and Plum-Pudding Molecules
Lec 12 - Overlap and Atom-Pair Bonds
Lec 13 - Overlap and Energy-Match
Lec 14 - Checking Hybridization Theory with XH_3
Lec 15 - Chemical Reactivity: SOMO, HOMO, and LUMO
Lec 16 - Recognizing Functional Groups
Lec 17 - Reaction Analogies and Carbonyl Reactivity
Lec 18 - Amide, Carboxylic Acid and Alkyl Lithium
Lec 19 - Oxygen and the Chemical Revolution (Beginning to 1789)
Lec 20 - Rise of the Atomic Theory (1790-1805)
Lec 21 - Berzelius to Liebig and Wöhler (1805-1832)
Lec 22 - Radical and Type Theories (1832-1850)
Lec 23 - Valence Theory and Constitutional Structure (1858)
Lec 24 - Determining Chemical Structure by Isomer Counting (1869)
Lec 25 - Models in 3D Space (1869-1877); Optical Isomers
Lec 26 - Van't Hoff's Tetrahedral Carbon and Chirality
Lec 27 - Communicating Molecular Structure in Diagrams and Words
Lec 28 - Stereochemical Nomenclature; Racemization and Resolution
Lec 29 - Preparing Single Enantiomers and the Mechanism of Optical Rotation
Lec 30 - Esomeprazole as an Example of Drug Testing and Usage
Lec 31 - Preparing Single Enantiomers and Conformational Energy
Lec 32 - Stereotopicity and Baeyer Strain Theory
Lec 33 - Conformational Energy and Molecular Mechanics
Lec 34 - Sharpless Oxidation Catalysts and the Conformation of Cycloalkanes
Lec 35 - Understanding Molecular Structure and Energy through Standard Bonds
Lec 36 - Bond Energies, the Boltzmann Factor and Entropy
Lec Last - Potential Energy Surfaces, Transition State Theory and Reaction Mechanism