Lec 34 - Physics 111: Rutherford Scattering (RUT)
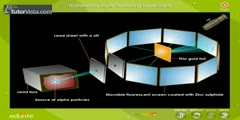
Physics 111: Rutherford Scattering (RUT) Physics 111 Advanced Laboratory. Professor Sumner Davis This video accompanies the Rutherford Scattering Experiment, providing students with an introduction to the theory, apparatus, and procedures. An experiment that shook the world of physics was carried out by Rutherford in 1910. He found that when scattering alpha particles from gold foils, far more of them were scattered at large angles than were predicted by theory. He explained the finding by assuming that all of the mass of an atom and positive charge of an atom are concentrated in a small volume at the center, in a compact nucleus. He derived an equation in which the differential cross section for scattering by an atomic nucleus is proportional to the inverse fourth power of the sine of the scattering angle. In this experiment the scattering formula is confirmed for the case of alpha particles scattered by gold nuclei in a thin gold foil. The charge on the gold nucleus will also be determined. http://advancedlab.org
Video is embedded from external source so embedding is not available.
Video is embedded from external source so download is not available.
Channels: Physics (General)
Tags: Physics 111 Rutherford Scattering RUT
Uploaded by: berkeleyphy111 ( Send Message ) on 19-09-2012.
Duration: 27m 50s
Here is the next lecture for this course
Lec 8 - Physics 111: Compton Scattering (COM)
20:27 | 4803 viewsLec 22 - Physics 111: Laser Induced Fluor ...
01:04:43 | 4793 viewsAlpha Scattering Experiment by Rutherford
01:30 | 8733 viewsRutherford's Experiment: Nuclear Atom
00:47 | 52455 viewsRutherford's Gold Foil Experiment
01:30 | 10678 viewsRutherford's Discovery Of The Nucleus
02:06 | 9422 viewsThe Standard Model of Particle Physics (5 ...
03:07 | 16688 viewsThe Standard Model of Particle Physics (P ...
03:07 | 17790 viewsQED -- The Jewel of Physics (12 of 15)
03:05 | 14012 viewsLec 1 - Modern Physics: Special Relativit ...
01:49:23 | 5833 viewsPhysics First with Dr. Leon Lederman
06:17 | 10350 viewsPhysics with Mr. Noon: Homework Help
07:39 | 7408 viewsPhysics with Mr. Noon: Snell's Law
09:53 | 10279 viewsPhysics with Mr. Noon: Reflection and Ref ...
09:44 | 14758 viewsPhysics with Mr. Noon: Light and Color
09:08 | 12181 viewsNo content is added to this lecture.
This video is a part of a lecture series from of berkeley
Lecture list for this course
Lec 1 - Physics 111: Atomic Physics (ATM) Part 1. Balmer Series
Lec 2 - Physics 111: Atomic Physics (ATM) Part 2. Zeeman Effect
Lec 3 - Physics 111: Beta Ray Spectroscopy (BRA)
Lec 4 - Physics 111: Brownian Motion in Cells (BMC)
Lec 5 - Physics 111: Instrumentation Section Lab Equipment (BSC)
Lec 6 - Physics 111: Bubble Chamber (BBC)
Lec 7 - Physics 111: Carbon Dioxide Laser (CO2)
Lec 8 - Physics 111: Compton Scattering (COM)
Lec 9 - Physics 111: Gamma Ray Spectroscopy (GMA)
Lec 10 - Physics 111: Hall Effect In A Plasma (HAL)
Lec 11 - Physics 111: Holography (HOL)
Lec 12 - Physics 111: Introduction to Error Analysis
Lec 13 - Physics 111: Josephson Junction Effect (JOS)
Lec 14 - Physics 111: Radiation and Laboratory Safety
Lec 15 - Physics 111: Laser Safety
Lec 16 - Physics 111: Atomic Physics (ATM) Theory Lecture ONLY
Lec 17 - Physics 111: Energy Levels Lecture Part 1
Lec 18 - Physics 111: Energy Levels Lecture Part 2
Lec 19 - Physics 111 Light Sources and Detectors Lecture
Lec 20 - Physics 111: Optical Instruments Lecture
Lec 21 - Physics 111: Energy Transitions Lecture Series
Lec 22 - Physics 111: Laser Induced Fluorescence and Raman Scattering (LIF)
Lec 23 - Physics 111: Low Light Signal Measurements (LLS)
Lec 24 - Physics 111: Non-Linear Spectroscopy and Magneto-Optics Part 1 (MNO)
Lec 25 - Physics 111: Non-Linear Spectroscopy and Magneto-Optics Part 2 (MNO)
Lec 26 - Physics 111: Atom Trapping (MOT)
Lec 27 - Physics 111: Muon Lifetime (MUO)
Lec 28 - Physics 111: Non-Linear Dynamics and Chaos (NLD)
Lec 29 - Physics 111: Nuclear Magnetic Resonance (NMR) Part-1 Continuous Wave
Lec 30 - Physics 111: Nuclear Magnetic Resonance Part-2 Pulsed NMR
Lec 31 - Physics 111: Optical Pumping (OPT)
Lec 32 - Physics 111: How to do an Oral Report
Lec 33 - Physics 111: Optical Trapping (OTZ)
Lec 35 - Physics 111: Hall Effect In A Semiconductor