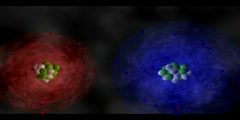
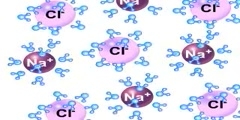
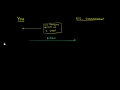
Ionic Bond: NaCl
Ionic bonds are a type of chemical bond based on electrostatic forces between two oppositely-charged ions. In ionic bond formation, a metal donates an electron, due to a low electronegativity to form a positive ion or cation. In ordinary table salt (NaCl), the bonds between the sodium and chloride ions are ionic bonds. Often ionic bonds form between metals and non-metals.
Channels: Scientific Animations Chemistry (General)
Tags: IonicBond cation anion salt
Uploaded by: sana ( Send Message ) on 15-05-2007.
Duration: 0m 20s