Nucleus of an atom
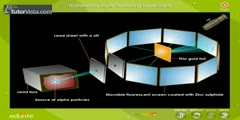
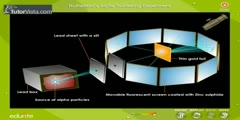
Nucleus of an atom was first discovered by deflection of positive particles passed through a thin foil of gold. The Geiger–Marsden experiment (also called the Gold foil experiment or the Rutherford experiment) was an experiment to probe the structure of the atom performed by Hans Geiger and Ernest Marsden in 1909,[1] under the direction of Ernest Rutherford at the Physical Laboratories of the University of Manchester. The unexpected results of the experiment demonstrated for the first time the existence of the atomic nucleus, leading to the downfall of the plum pudding model of the atom, and the development of the Rutherford (or planetary) model. A beam of alpha particles, generated by the radioactive decay of radium, was directed normally onto a sheet of very thin gold foil. The gold foil was surrounded by a circular sheet of zinc sulfide (ZnS) which was used as a detector: the ZnS sheet would light up when hit with alpha particles. Under the prevailing plum pudding model, the alpha particles should all have been deflected by, at most, a few degrees; measuring the pattern of scattered particles was expected to provide information about the distribution of charge within the atom. However they observed that a very small percentage of particles were deflected through angles much larger than 90 degrees. According to Rutherford: ''It was quite the most incredible event that has ever happened to me in my life. It was almost as incredible as if you fired a 15-inch shell at a piece of tissue paper and it came back and hit you. On consideration, I realized that this scattering backward must be the result of a single collision, and when I made calculations I saw that it was impossible to get anything of that order of magnitude unless you took a system in
Channels: Experiments Chemistry (General) Analytical chemistry
Tags: nucleus of an atom deflection thin foil
Uploaded by: jamvj ( Send Message ) on 07-06-2009.
Duration: 0m 48s