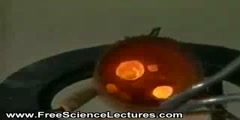
A home video showing Endothermic Reaction
In thermodynamics, the word endothermic ("within-heating") describes a process or reaction in which the system absorbs energy from the surroundings in the form of heat. Its etymology stems from the Greek prefix endo-, meaning “inside” and the Greek suffix –ther, meaning “heat”. The opposite of an endothermic process is an exothermic process, one that releases energy in the form of heat. The term endothermic was coined by Marcellin Berthelot (25 October 1827 – 18 March 1907). The concept is frequently applied in physical sciences to, for example, chemical reactions, where thermal energy (heat) is converted to chemical bond energy.
Channels: Chemistry (General)
Tags: Endothermic Reaction
Uploaded by: buraktube ( Send Message ) on 09-11-2010.
Duration: 0m 50s