How antioxidants work
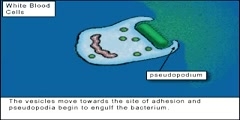
Everything from tea to carrots to vegetable oils are being promoted as being good for your health. This is because these and many other foods contain antioxidants. Vitamin A, C and E (alpha-tocopherol) capsules are being consumed in larger and larger quantities because of their well established antioxidant properties. Oxidation of a wide range of chemical compounds and the production of radicals at the cellular level are believed to be involved in the cause of many cancers and may also be important factors causing cardiovascular disease. This is why there is so much interest in antioxidants. But how exactly do antioxidants work? The key role played by antioxidants in the body is their ability to react with radicals. When this happens the destructive properties of the radical is eliminated. A free radical is a chemical compound that contains one or more unpaired electrons. Radicals can be produced by exposure to energy such as radiation or may be the product of incomplete reactions in the cells that produce electrons that have escaped . The step that produces a radical is called the initiation step. Radicals are usually represented in chemical formula by a single dot. Below are three examples of initiation steps that illustrate the production of radicals.
Channels: Medical
Tags: How antioxidants work
Uploaded by: buraktube ( Send Message ) on 09-11-2010.
Duration: 2m 40s