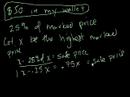Lec 31 - The Two Ozone Problems
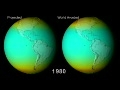
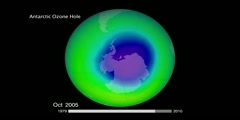
"Lec 31 - The Two Ozone Problems" The Atmosphere, the Ocean and Environmental Change (GG 140) There are two ozone problems in the atmosphere. Tropospheric ozone in the form of photochemical smog is sometimes dangerously high whereas stratospheric ozone concentration is sometimes dangerously low. Photochemical smog is created through chemical reactions between UV radiation from the run and nitrogen oxides that are emitted from automobiles. High concentrations of tropospheric ozone are dangerous because of the damage ozone can cause to a person's airway if it is inhaled. The EPA has specified limits of ozone concentration but several counties in the USA exceed these limits. The primary air pollutants from which ozone is created have a peak concentration twice a day typically, which is associated with rush hour times during the day. 00:00 - Chapter 1. Air Pollutants 06:05 - Chapter 2. Two Ozone Problems 07:08 - Chapter 3. Ozone Properties 11:20 - Chapter 4. Photochemical Smog 15:16 - Chapter 5. Ozone Concentration Limits 25:10 - Chapter 6. Creation of Ozone Pollution 26:41 - Chapter 7. Primary Pollutants Complete course materials are available at the Open Yale Courses website: http://oyc.yale.edu This course was recorded in Fall 2011.
Video is embedded from external source so embedding is not available.
Video is embedded from external source so download is not available.
Channels: Environmental engineering
Tags: Lec 31 - The Two Ozone Problems
Uploaded by: yaleatmoocenchng ( Send Message ) on 13-09-2012.
Duration: 37m 51s
Here is the next lecture for this course
Solving Word Problems
07:12 | 7956 viewsAge word problems 2
09:56 | 6874 viewsAge word problems 3
04:08 | 7922 viewsDepletion of ozone layer
04:54 | 17300 viewsOzone Layer
02:00 | 7081 viewsHow ozone layer is reduced
02:20 | 5405 viewsOzone mapper
03:38 | 4113 viewsDental problems diagnosis
01:54 | 5506 viewsNew Surgery Methods Help Patients with Sp ...
03:20 | 4542 viewsMathematical Problems For Geometry
01:01 | 5223 viewsLec 107 - SMOG OR JOBS? The Impact of Tig ...
01:07:45 | 3575 viewsLec 53 - Introduction to rate-of-change p ...
09:42 | 4018 viewsLec 18 - Trigonometry word problems (part 1)
09:56 | 4326 viewsLec 19 - Trigonometry word problems (part 2)
02:51 | 3970 viewsLec 23 - More percent problems
09:05 | 3551 viewsNo content is added to this lecture.
Lecture list for this course
Lec 1 - Introduction to Atmospheres
Lec 2 - Retaining an Atmosphere
Lec 4 - Vertical Structure of the atmosphere; Residence Time
Lec 5 - Earth Systems Analysis (Tank Experiment)
Lec 6 - Greenhouse Effect, Habitability
Lec 9 - Water in the Atmosphere I
Lec 10 - Water in the Atmosphere II
Lec 11 - Clouds and Precipitation (cloud chamber experiment)
Lec 12 - Circulation of the Atmosphere (Exam I review)
Lec 13 - Global Climate and the Coriolis Force
Lec 14 - Coriolis Force and Storms
Lec 18 - Seasons and Climate Classification
Lec 19 - Ocean Bathymetry and Water Properties
Lec 20 - Ocean Water Density and Atmospheric Forcing
Lec 22 - Ocean Currents and Productivity
Lec 24 - Ice in the Climate System
Lec 25 - Ice and climate change
Lec 26 - Isotope Evidence for Climate Change
Lec 28 - Global Warming (continued)
Lec 29 - Global Warming (continued)
Lec 30 - Climate Sensitivity and Human Population