Chemical Science - Chemical Equilibrium (cont.) - Lecture 20
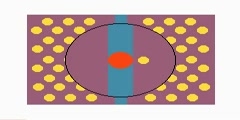
Principles of Chemical Science/n * Email this page/nVideo Lectures - Lecture 20/nTopics covered: /nChemical Equilibrium (cont.)/nInstructor: /nProf. Catherine Drennan/nTranscript - Lecture 20/nToday we are going to continue with chemical equilibrium./nAnd at the end of the lecture, Professor Ceyer will be here to talk about exam two. We are going to start off where we left off, and we were talking about Le Châteier's principle. Le Châteier had this great idea that systems tend to respond to stress in a particular way./nThat if you apply a stress to a system at equilibrium, it will respond to that stress by trying to minimize the stress. The reaction will respond to adjust for the stress that was applied and minimize that stress. If you consider what stress is applied to a reaction and then how that stress can be minimized, you can predict whether the reaction will go in the forward direction or the reverse direction./nAnd so it tends to go in one of those directions to minimize the stress until the stress had been minimized and then equilibrium is reached again. We talked last class about adding and removing products or reactants and how the system would respond. And today we are going to consider changes in volume to a gaseous system and also temperature and how systems respond./nAnd at the end we will talk about the relevance of this to biological systems. Again, here is Le Châteier. We can use this to predict the direction of the reaction, the response to the stress that is applied. First let's consider what happens if you have a mixture of gases at equilibrium. They are reacting. Again, in equilibrium, it is a dynamic process./nYou have the reaction going in the forward direction and reverse direction, but the rates of those reactions are the same so there is no net change in composition but the reaction is still happening. Now, if you have a system at equilibrium, a gaseous system in equilibrium and you change the volume, it will consider how the reaction will shift to compensate for this stress./nSo a decrease in volume of the gaseous system will cause an increase in total pressure. How do we know that? What equation tells us about pressures and volumes of gases? Yeah, ideal gas law, PV equals NRT. That is what we will be considering today. Le Châteier's principle would predict that the system will respond in such a way then to reduce the total pressure./nIf you decrease the volume, increase the total pressure, the system is going to respond then to reduce that total pressure, to minimize that stress that was added into the system./nLet's look at an example. Here we have 2P2 gas going to P4 gas, so two molecules of P2 going to one of P4. And in the middle one here, we have this system at equilibrium. And we are going to consider both what happens when we decrease the volume and when we increase the volume. First let's consider this decrease./nWhat happens is that a decrease in volume shifts the reaction toward products. Why is that true? Well, it has to do with the stoichiometry of this equation. For every two moles here that are consumed, only one is formed. A shift to the right, toward products is going to reduce the number of molecules in the system, and therefore reduce the total pressure./nWhen you have a decrease in volume that initially causes the total pressure to increase. And so the system responds in such a way to minimize that stress and decrease the pressure, which you can do by shifting this reaction toward products or to the right. This is what Le Châteier's principle would allow you to guess when you look at this particular reaction./nAnd you can also prove to yourself that this is correct by considering Q and K and delta G. Let's prove that this is what would, in fact, happen. We consider again Q and K. Remember that this is what is important in both of these two lectures. We are talking about Q, the reaction quotient, the ratio of products to reactants under whatever condition you are at that particular moment in time./nWhereas, K is our equilibrium constant, the ratio of products to reactions at equilibrium. Now to make the math easy, let's assume that the volume decrease happens by a factor of two. You decrease the volume by two. That is going to initially increase the pressure of both gases of P2, the reactant, and P4, the product by two, so the partial pressure is going to go up./nNow we can figure out by how much with this decrease in volume. Q, again, the reaction quotient, is products over reactants, so our product, partial pressure of P4 over the partial pressure of P2 raised to the two. Again, you consider the stoichiometry when you are writing your equation for Q or for K./nIf we work this out, we find that by decreasing the volume by a factor of two, Q is now half what it was. It is decreased by a factor of two. Now Q is less than K. We were at equilibrium before, but if stress was implied the volume was decreased. And now we have a situation where Q is no longer equal to K, Q is less than K. And so that is going to then shift the reaction toward products./nIt will be spontaneous in the forward direction. You can just sort of rationalize what will happen and you will have questions on homework and on exams saying if this stress is applied, predict how the reaction is going to respond, you can work it out in terms of thinking about Q and K or just sort of rationalize how do you minimize that stress that has been applied to the system?/nAnd if you are asked how much happens if numbers are given, you can actually work this out, what Q is at that point and what K was. Now, if we increase volume, if we go in this direction, what is going to happen? Well, the reverse will happen in this case. We are going to shift toward reactants./nFor every one mole of product that goes in this direction, in the reverse direction you will form these two moles of P2. A shift toward the reactants, a shift to the left is going to increase the total pressure. If you increase the volume, you are initially going to decrease the partial pressure. And a shift in this direction of the reaction will compensate, it will increase that partial pressure until you reach equilibrium again./nBecause you are going in a reverse direction from one to two moles. Again, we could work this out in Q and K. Actually, we will do this at a later example. But, again, you can rationalize or figure it out in terms of Q and K and what delta G would be, positive or negative, which direction the reaction is spontaneous, in the forward direction or in the reverse direction. All right./nLet's make this a little bit more complicated now and consider what happens when you add an inert gas into that mixture. Here we are adding an inert gas into the container. And when we add it, it is increasing the total pressure of that container and temperature is constant. Add an inert gas. Increase the total pressure. What is going to happen to the reaction? Will it shift to the left or the right?/nWhat is going to happen? The answer is in this case nothing. Why? Why won't this affect the equilibrium? Why aren't we affecting Q here? The answer is that Q depends on the partial pressures of the gases involved and partial pressures are not changing. The total pressure is changing./nBut the partial pressures of those gases are not changing. At this point, let's review partial pressure, and then I think this will make a lot of sense. The definition of partial pressure is the pressure that each gas would exert if it were alone in the container. Here we have some oxygen at one atmosphere, here we have nitrogen at one atmosphere, and here we have a mixture of the oxygen and the nitrogen./nAnd now that we have added one gas to the other, the pressure is two atmospheres. We added one atmosphere of this and one atmosphere of that, and we have two atmospheres total. But if you are asking about the partial pressure, say, of oxygen, the partial pressure of oxygen is the same here as it in the mixture because partial pressure is the pressure that is due just to that particular gas, as if it were alone in a container./nLet's look at the equations for partial pressure. And, if you haven't had partial pressure before in high school, I said this is a review, but this is actually everything you need to know about partial pressure for this unit so it doesn't really matter if you haven't seen this before. If we have our familiar ideal gas law, the partial pressure of A depends on just the number of molecules of A in that container at a given volume./nThe total pressure of a system will be the partial pressure of A plus B plus C, depending on how much of a mixture you have. In this case, we have a mixture of 02 and N2, so our total pressure is two. But our partial pressure of each of these gases in here is still one, as it was before when they were alone./nHow is this all relevant? Well, it is relevant because Q depends on the partial pressure, not the total pressure. The question you have to ask yourself in looking at all these problems is, is the partial pressure changing, i.e., is Q changing? Because only when Q changes will equilibrium be affected. In the particular example that I gave you at the beginning here, I asked what happens if an inert gas is added to a container increasing the total pressure at constant temperature?/nAnd so the answer was nothing because Q is not affected. The partial pressure for the gases is still the same. The volume has not changed any. You have not added more of these two gases. You added an inert gas so the number of moles of these are the same. The volume is the same so the partial pressure is the same. And if partial pressures are the same then Q is unaffected so Q does not change./nIt doesn't matter. There is no shift because Q is still equal to K. We are still under these conditions. It seems like it is a stress applied, but it is not really a real stress so there is no change. Let's look at another scenario and consider what will happen. Now what happens if we add an inert gas but the total pressure and the temperature are kept constant?/nThe total pressure is not allowed to change, it is kept constant. For this to be true, for you to add two things together and the total pressure doesn't change, what had to have happened? Volume had to have changed here, otherwise this couldn't be true. The volume had to change./nBecause here we have your constant temperature, we have the gas constant here, number of moles. If we have increased this number of moles and then total pressure has to be affected by it. If it didn't that means volume had to have changed for that to be true. For the pressure to be constant, the volume of the container must have increased. And so you will notice, in working a lot of these problems, often it won't say anything about the volume./nIt will tell you about the pressure, and you have to figure out whether that means there was a volume change because the volume changes are the ones that are relevant. It is all sort of in the wordings of these problems. All right. The volume must have increased otherwise the pressure would have gone up. Then this question really is what happens when the volume increases? And we know how to answer that question already./nFor this particular reaction, where you have these two P2s going to one P4 and increase in volume means that you are going to shift two reactants, you are going to go from this side that has one to this side which has two. That way, when you increase the volume, the partial pressure is going to drop. You want to respond in a way to minimize that stress, which means you want then an increase in the partial pressure./nAgain, we can think about this in terms of Q and K. Then we just work a little example on the board to emphasize the point. We did the other example earlier, but now let's consider if your volume doubles. And my picture doesn't make it look like it is doubling but this makes the math easier. That means if the volume doubles you are going to half the partial pressure initially of both gases, of P2 and P4./nAnd so now we consider Q because it is really all about the relationship between Q and K. Q is going to equal the partial pressure of the products over reactants, and the reactant is partial pressure of P2. Again, with the stoichiometry that is squared. If the volume doubles, initially the partial pressure will be half for the products and also half for the reactant P2, and that is squared./nSo, Q is twice what it was before. Now Q is greater than K. And so you will have a delta G that is positive, which means you are going to be spontaneous in the direction toward reactants./nAnd you could sort of just reason that out by looking at the number of moles on one side or the other. Or, if you want to convince yourself, you can look at Q and K because this is really where it comes from. When Q equals K, you are at equilibrium. If it is greater than K then you shift. Question? Yeah, just because it doubles./nThere could be real numbers that you are looking at, but this is just considering if you double this then you are going to have a decrease. Yeah, it's a partial pressure. The number of moles are not changing but the partial pressure is changing because the volume is changing. All right. Yeah?/nWell, at equilibrium Q is equal to K. That Q and K are both products over our reactants or the partial pressure of products over reactants. When you make some change like this Q was equal to K, but now you have a change. Either if you increase or decrease then it changes the relationship to your equilibrium so you are no longer in the equilibrium conditions./nAt equilibrium Q equals K by definition./nYou are always considering, if you are talking about applying a stress to a system, you are assuming that the system was in equilibrium. And then you apply a stress to that situation that is at equilibrium and ask how does this system respond? In all of these questions you are under equilibrium conditions and then you apply the stress./nSo that's volume. And so the secret to these problems is asking is the partial pressure changing under those conditions when you are talking about volume? You had a system at equilibrium. Then you messed up your system at equilibrium by compressing the container or increasing the container./nAnd whether you added an inert gas, it really always falls down to those two possibilities that the volume was decreased or the volume was increased. You have to look at the question and read carefully and say what is happening to the volume under these cases, and then you can figure out which direction it is going to go. And, of course, that was all one example. You have to look at what the particular reaction is, how many moles are on one side and how many are on the other to think about this./nLe Châteier also works for temperature. In this case, you are thinking about what happens if you raise the temperature of an equilibrium mixture. The reaction mixture is at equilibrium, delta G equals zero, Q equals K and now you are changing something. You are changing the temperature. Raising the temperature by adding heat will cause the reaction to shift so that that heat is absorbed./nThis is the same idea. Applying stress to the system, raising the temperature, the reaction is going to respond so that the heat that was added is now absorbed. You go back to where you started. Le Châteier works for temperature as well, it is the same idea, but here the stress is changing the temperature of the system./nAdding heat or removing heat from the system and thinking about how that reaction that was at equilibrium is going to respond to this stress. Let's consider an exothermic reaction first. An exothermic reaction, if we are raising the temperature of an exothermic reaction you will favor the formation of the reactants./nWhy is that true? Well, with an exothermic reaction you are producing heat. And in the reverse direction you would be absorbing heat. According to Le Châteier's principle you want to respond to minimize the stress. If you raise temperature you want to shift in a direction where that heat will be absorbed./nSo a shift then in the endothermic direction. And so for an endothermic reaction, it is just the reverse of this. For an endothermic reaction, you are absorbing heat toward products and producing heat toward reactants. If you raise the temperature of an endothermic reaction, you will favor the formation of products. Because, again, if you are adding heat, you want to absorb the heat so it will shift the reaction in a direction that absorbs the heat./nLet's consider this a bit further. In this case, delta H is our predictive tool. If we know delta H, we are going to be able to predict the direction that the reaction is going to go. Before we were talking about delta G as being our indicator. Here delta H is telling us quite a bit about what is going to happen, i.e., whether the reaction is exothermic or endothermic./nLet's just do a quick example. Here is a reaction, delta H negative 197.78 kilojoules per mole. Is that an endothermic or exothermic reaction? Exothermic reaction. If heat is added, if the temperature is raised, what direction will it go, toward products or reactants?/nReactants. It will want to absorb that heat, so it goes in the endothermic direction or the reverse direction. That is pretty simple. Delta H allows us to make the prediction. All right. We need to think now about the relationship between temperature and equilibrium constants./nEquilibrium constant has been constant up until this point, but now we see that temperature will change the equilibrium constant. The ratio of products to reactants that you have at equilibrium will depend on whether it is an exothermic, endothermic reaction and how much heat is in the system, the temperature that the reaction is being run at. You will get a different equilibrium constant, a different ratio of products to reactants if you are at one temperature versus another temperature./nAnd also, we will come back to this at the end of the course, reaction rates depend on temperature. How fast those reactions go depends on temperature as well. All right. Now we want to figure out exactly how equilibrium constants depend on temperature./nAnd to do that we have to think about what we know in terms of equations that have temperature in them and have equilibrium constants in them. We know that delta G knot is minus RT natural log of K. We also know that delta G knot is equal to delta H knot minus T delta S. Now, if we want to take that information and combine it and solve for the equilibrium constant in terms of delta H --/nAnd we want to do that. We want to solve for in terms of delta H and in terms of temperature, because we want to think about delta H, endothermic, exothermic reaction and the relationship between equilibrium constant and temperature. If we just rearrange these equations that you have already seen and should be reviewing now for exam two, you can come up with this. Now we want to think about which of these terms are temperature dependent./nI have already told you that the equilibrium constant is temperature dependent. Delta H here and delta S, we could assume that they are pretty much constant over any temperature that we would be looking at, any temperature that a reaction would be run. These guys are not really changing. And so if they are not really changing then we can see that the equilibrium constant is going to change with respect to the change in temperature./nAs we change temperature, K will vary here. And we can look at this for two different temperatures. If you change the temperature of the system, the first temperature and the second temperature, T1 and T2, they will be different equilibrium constants at these two temperatures, different ratios of products, two reactants at these different temperatures./nIf we write out two equations then, one for K2 with T2, so the equation for the second temperature and the equation for the first temperature at T1 and the first equilibrium constant for that particular temperature. And then if we subtract these two equations, so if we subtract the natural logs of the Ks, we can express that in terms of natural log of K2 over K1./nAnd if we subtract the second part, we subtract this from this, the delta S over R terms are going to cancel out, they are the same, delta S is not changing with temperature here, and so those two terms would be the same, they are going to go away. And now what is different are these two temperatures. We can pull out the negative delta H here./nWe can pull out the one over R. And that leaves one over T2 minus one over T1. And it has a name so you know this must be an important equation. That is partly how you can tell. It is the van't Hoff equation. It tells you how the equilibrium constants are going to change with temperature./nNow let's look at how the numbers are going to work out if we have a negative sign here, i.e., if it is an exothermic reaction, or we are going to have a positive delta H, so that would be an endothermic reaction, and whether you are adding or removing heat. There will be a difference in the ratios of K1 to K2. Let's think about what comes out of this equation and rationalize it./nThis looks a little complicated but it is actually pretty simple. Here all we are doing is saying what if delta H is negative? If delta H is negative and we have T2 greater than T1, well, this negative sign here is going to be negative all the way through here. It is always present. That is part of the equation. Delta H now, if it is negative, you will have a negative here./nAnd then we consider the different between one over T2 and one over T1. And, in this case, if T2 is greater than T1, that is a negative. When you multiplied all those out together we would have a negative here. That is just what is all done there. Now let's rationalize that this makes sense. This is just the same thing but has a little more rationalization in it. It puts things in words instead of using the greater and less than thing./nAnd these parts are not written in your notes, but if you want to add the words to it, that is fine, on the handout. And you can just think about this on your own as well. If delta H is less than zero, negative number, you have an exothermic reaction. What happens if you increase the temperature of an exothermic reaction? Let me just put those back up on the board for a minute. For an exothermic reaction, we are talking about reactants going to products. In an exothermic reaction heat is produced in the forward direction and heat is absorbed in the reverse direction./nIf we increase the temperature, it is going to want to go in a direction to compensate for that to absorb the heat, so it is going to go toward reactants. And so you are going to have a K1 that is greater than K2, which means at the new temperature you are going to have less products or more reactants. And we can rationalize why that would be true. And, if you don't want to rationalize it, you can always take the van't Hoff equation and figure it out by looking at the different signs and what it would mean./nWhat is going to happen in terms of that ratio of K1 to K2 when you have this particular condition? Now, if you decrease the temperature of an exothermic reaction then it wants to compensate by producing more heat. You decrease the temperature. It wants to respond to the stress to minimize the stress. The way to minimize the stress is produce more heat so you will make more products./nNow for the endothermic reaction, it is just the other way around./nReactants going to products. Here heat is absorbed. And in the reverse direction heat is produced./nNow, if we have our endothermic reaction and we increase temperature, we are going to shift to our product because we are going to want to absorb that heat that was just added, minimize the stress. And, for an endothermic reaction, if we decrease the temperature then we are going to shift it to produce more heat, i.e., toward reactants, so we will have less products. And we would have K1 that is greater than K2./nYou can use the van't Hoff equation to rationalize the prediction of change, or, if you are given numbers, you can say the exact amount of change. If you are given a K value at one temperature and said if we change this temperature, if we increase it, this is what delta H is, what is our new equilibrium constant? You can use the van't Hoff equation to come up with direct numbers or you can use it to just rationalize the direction of change./nIf you have less time on a test, you can also just think about what might be going on here and predict without working through the numbers. Now we are going to apply what we just learned to think about how it can be used./nYou want to think about the shifts that we talked about last class and the ones today to have options of adding and removing products or reactants, changing volumes, changing temperatures. This is quite an important reaction, and we talked about this last time, nitrogen gas plus hydrogen gas going to ammonia. It is an exothermic reaction./nIf it is exothermic and you want to drive it toward ammonia, which is what people do. They want to make ammonia and want what is called fixed nitrogen. Ammonia is used in fertilizers, lots of nitrogen in the air, but you don't want it in the air, you want it in your soil for growing better crops, so you want to do this reaction. You want to maximize the yield. Then it seems like the low temperature would be good. It's an exothermic reaction so low temperature would be good. It would favor product./nBut there is a problem with that, which is that low temperature also slows the rate. And you usually want a good rate. You want to make a lot of products pretty quickly, make some money off of it, so you have to balance here. Sometimes the thermodynamics and the kinetics are working against each other. What would favor one doesn't work so well for the other. They have to have a compromised temperature, which is actually quite a high temperature of 500 degrees Celsius./nThere is a little bit of a problem there. The temperature thing isn't working for us. What are some other things that they could do to drive the reaction toward products? What else could you do? OK. What else could you do?/nI am hearing a couple of different things. I am looking for one other. What about --/nA couple things that you could do that would work, I think I heard most of them now. Remove products as they are formed. If you remove this product that would drive it toward more products. And so one thing that they actually do is every once in a while stop the reaction, cool it all down and liquefy the ammonia gas./nThe liquid pours off and then you remove this product and then you make more product. If it reaches equilibrium, you pull off the product and then it makes more. And I also heard about this compressing the volume of the container because over here the reactants, there are four molecules that are reacting to form two, and so that should also shift in the correct direction to make product./nThey actually do these at this pretty high temperature, and they have to apply a really high pressure to compress that volume. This is expensive. And it requires a lot of energy, actually, to do this reaction because of the conditions that are required to maximize the yield. Again, why this is important, there is a lot of nitrogen in the air, however, it is really stable. And you could probably tell me why it is so stable if you draw a nice Lewis diagram of it./nIt is difficult to break these apart, so that is why this is a hard problem. Because you want to break this bond to have usable nitrogen and it is challenging to do. The process, which I just described to you, is one that was developed quite a long time ago. This reaction is called the Haber-Bosch process and is responsible for generating 1.6 times 10 to the 10 kilograms of ammonia each year in the U.S. for fertilizers and other things./nAnd it was developed by these two men right here. Haber came up with the initial idea, and Bosh helped figure out the appropriate conditions to get the maximum yield. All they are doing is basically using some of the principles that you have already learned in this course. And they both won Nobel Prizes for being part of this process./nThis one initially and this one later on. I will also mention that this is an interesting man if you are interested in sort of history of science. He is a German chemist, and during World War I he switched his effort from fertilizers to coming up with different kinds of poison gases that could be used in warfare. But he was not rewarded favorably for this later on because he was also Jewish./nIn 1933, he had to flee Germany, despite all that he had done for the war effort, earlier on. He did not live too long a life after that. But this was developed a lot time ago and is still used. Some scientists are actually trying to figure out how to do a better job. They figure any process that really started to be developed in the 1800s and kind of refined in the 1900s, maybe we could do better. Again, a lot of energy is responsible, high temperatures, these extreme pressures for the condensed volume./nMaybe we could figure out a way to do that same reaction without those extreme conditions that require a lot of energy. And so a number of scientists have looked to an enzyme, a protein found in bacteria that does the exact same reaction as what is done commercially but doesn't use any high temperatures or any high pressures. A number of scientists are trying to figure out how this protein works./nHow it is able to do this reaction so efficiently. Here is a space filling picture of the enzyme, and here are the tallow clusters that do the actual chemistry. And so orange is iron and yellow is sulfur. Blue is nitrogen. Red is oxygen. Gray is carbon. This purple color here is molybdenum. These are metals that are used in biology to do particular kinds of reactions./nAnd this just shows a little bit more about the cluster sort of in the heart of this protein molecule. And all the chemistry happens at the metals, but it is surrounded by a lot of protein. That is one effort that a number of scientists are venturing on. Often we find varied forms of bacteria have proteins that do reactions that we want to do, and they do it much better than we do them. And so we want to understand how that works./nAnd this is one example. It turns out this is quite complicated, and scientists still don't understand the exact details of the reaction mechanism at that metal site. Another attempt is just to pretend to move away from nature. And Dick Schrock's lab here at MIT has been able to do this reaction using a sort of designed catalyst that has molybdenum in the center. You have probably heard the name Dick Schrock because he won the Nobel Prize in chemistry just a few weeks ago./nHe didn't win for this. This is another one of his contributions. All right. Really quickly, I want to give this one more biological example. And then we will hear more about exam two. Again, as I will emphasize in this course. All the things you learn can be applied to biological systems as well. The same basic principles./nHere we have hemoglobin. Hemoglobin is the protein in your body that carries oxygen from the lungs to the cells. And you can write the reaction for hemoglobin and oxygen the way you can write any of the reactions we have written. Hemoglobin plus oxygen goes to oxyhemoglobin. Here this is just oxygen bound to the hemoglobin. Now let's consider what happens if you decide to climb a mountain./nThe partial pressure of oxygen is different at sea level than it is in the mountains. It is higher. As you go up higher altitude, the partial pressure of the oxygen decreases. What does this mean in terms of this equilibrium? Which direction would it shift if the partial pressure of this is decreased?/nYeah, you are going to shift toward reactants, which means you have less oxygenated blood, you have less oxygen bound. And that is not good. It makes it much harder to get energy to climb that mount, so that is not good. Usually, if you are going to go climb a mountain, you have to adjust, you have to acclimatize. How many people are mountain climbers?/nAnybody? A few people. You usually have to get up and wait for a while before you can start climbing. What actually happens is your body is making more hemoglobin, so you have to wait for this to happen. And, if it makes more hemoglobin, what direction does it go? Right, it shifts toward products. Then you are in better shape to climb your mountain. Again, the same principles of shifting reaction applying to a biological system.
Channels: Chemistry (General)
Tags: Chemical Equilibrium
Uploaded by: mitlectures ( Send Message ) on 16-04-2009.
Duration: 44m 32s