Course: Chemistry Dnatube
share this page with the world.

Lec 1 - Elements and Atoms
Elements and Atoms How elements relate to atoms. The basics of how protons, electrons and neutrons make up an atom.
Lec 2 - Introduction to the atom
Introduction to the atom The atom, proton, neutron and electron
Lec 3 - Orbitals
Orbitals An introduction to orbitals
Lec 4 - More on orbitals and electron co ...
More on orbitals and electron configuration More intuition on orbitals. Touching on electron configuration.
Lec 5 - Electron Configurations
Electron Configurations Introduction to using the periodic table to determine electron configurations
Lec 6 - Electron Configurations 2
Electron Configurations 2 Figuring out configurations for the d-block elements
Lec 7 - Valence Electrons
Valence Electrons Looking at valence electrons to figure out reactivity
Lec 8 - Groups of the Periodic Table
Groups of the Periodic Table Properties of alkali, alkaline earth and transition metals. Halogens and noble gases.
Lec 9 - Periodic Table Trends: Ionizatio ...
Periodic Table Trends: Ionization Energy What an ion is. Using the periodic table to understand how difficult it is to ionize an atom.
Lec 10 - Other Periodic Table Trends
Other Periodic Table Trends Electronegativity, metallic nature and atomic radius.
Lec 11 - Ionic, Covalent, and Metallic Bonds
Ionic, Covalent, and Metallic Bonds Introduction to ionic, covalent, polar covalent and metallic bonds.
Lec 12 - Molecular and Empirical Formulas
Molecular and Empirical Formulas Introduction to molecular and empirical formulas. Calculating molecular mass.
Lec 13 - The Mole and Avogadro's Number
The Mole and Avogadro's Number Introduction to the idea of a mole as a number (vs. an animal)
Lec 14 - Formula from Mass Composition
Formula from Mass Composition Figuring out the empirical formula from a molecules mass composition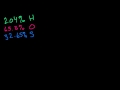
Lec 15 - Another mass composition problem
Another mass composition problem Another exercise converting a mass composition to an empirical formula.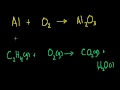
Lec 16 - Balancing Chemical Equations
Balancing Chemical Equations The art of balancing equations in chemistry!
Lec 17 - Stoichiometry
Stoichiometry Introduction to stoichiometry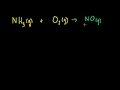
Lec 18 - Stoichiometry: Limiting Reagent
Stoichiometry: Limiting Reagent Stoichiometry problem where we have a limiting reagent!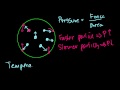
Lec 19 - Ideal Gas Equation: PV=nRT
Ideal Gas Equation: PV=nRT Intuition behind the ideal gas equation: PV=nRT.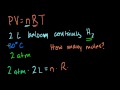
Lec 20 - Ideal Gas Equation Example 1
Ideal Gas Equation Example 1 Figuring out the number of moles of gas we have using the ideal gas equation: PV=nRT.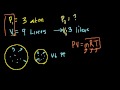
Lec 21 - Ideal Gas Equation Example 2
Ideal Gas Equation Example 2 PV/T is a constant. Figuring out the volume of an ideal gas at standard temperature and pressure (STP).
Lec 22 - Ideal Gas Equation Example 3
Ideal Gas Equation Example 3 Figuring out the mass of Oxygen we have.
Lec 23 - Ideal Gas Example 4
Ideal Gas Example 4 Figuring out the molar mass of a mystery molecule at STP.
Lec 24 - Partial Pressure
Partial Pressure Figuring out the partial pressures of various gases in a container.
Lec 25 - States of Matter
States of Matter Introduction to the states or phases of matter.
Lec 26 - States of Matter Follow-Up
States of Matter Follow-Up More on Plasma and Hydrogen bonds.
Lec 27 - Specific Heat, Heat of Fusion a ...
Specific Heat, Heat of Fusion and Vaporization Specifict heat and phase changes: Calculating how much heat is needed to convert 200g of ice at -10C to 110 degree steam.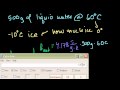
Lec 28 - Chilling Water Problem
Chilling Water Problem How much ice at -10 degrees C is necessary to get 500g of water down to 0 degrees C?
Lec 29 - Phase Diagrams
Phase Diagrams Understanding and interpreting phase diagrams
Lec 30 - Van Der Waals Forces
Van Der Waals Forces Van Der Waals Forces: London Dispersion Forces, Dipole Attractions, and Hydrogen Bonds.
Lec 31 - Covalent Networks, Metallic, an ...
Covalent Networks, Metallic, and Ionic Crystals Covalent Networks, Metallic, and Ionic Crystals: Some of the strongest molecular structures.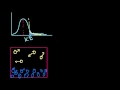
Lec 32 - Vapor Pressure
Vapor Pressure Vapor Pressure, Volatility, and Evaporation
Lec 33 - Suspensions, Colloids and Solutions
Suspensions, Colloids and Solutions Suspensions, Colloids and Solutions. The difference between Molarity and Molality.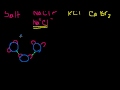
Lec 34 - Solubility
Solubility Solubility of salt and gas solutes in liquid solvent.
Lec 35 - Boiling Point Elevation and Fre ...
Boiling Point Elevation and Freezing Point Supression Raising or lowering the boiling or freezing point of a solution by adding solute.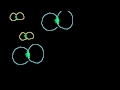
Lec 36 - Introduction to Kinetics
Introduction to Kinetics Kinetics, activation energy, activated complex and catalysts.
Lec 37 - Reactions in Equilibrium
Reactions in Equilibrium Equilibrium reactions and constants.
Lec 38 - Mini-Video on Ion Size
Mini-Video on Ion Size Correcting a mistake and learning a bit about ion size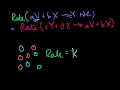
Lec 39 - Keq Intuition (mathy and not ne ...
Keq Intuition (mathy and not necessary to progress) A probabilistic look at how molecules react to develop the intuition behind the equilibrium constant formula.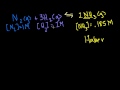
Lec 40 - Keq derivation intuition (can s ...
Keq derivation intuition (can skip; bit mathy) A more concrete attempt at showing how the probabilities of molecules reacting is related to their concentration.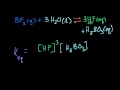
Lec 41 - Heterogenous Equilibrium
Heterogenous Equilibrium Ignoring the solution or the solid state molecules when calculating the equilibrium constant.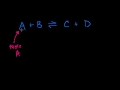
Lec 42 - Le Chatelier's Principle
Le Chatelier's Principle Le Chatelier's Principle regarding the "stressing" of reactions in equilibrium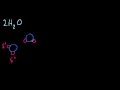
Lec 43 - Introduction to pH, pOH, and pKw
Introduction to pH, pOH, and pKw Autoionization of water into hydronium and hydroxide ions. pH, pOH, and pKa.
Lec 44 - Acid Base Introduction
Acid Base Introduction Arrhenius, Bronsted Lowry, and Lewis Acids and Bases.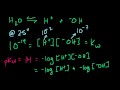
Lec 45 - pH, pOH of Strong Acids and Bases
pH, pOH of Strong Acids and Bases Calculating the pH or pOH of strong acids and bases.
Lec 46 - pH of a Weak Acid
pH of a Weak Acid Calculating the pH of a weak acid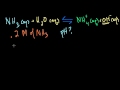
Lec 47 - pH of a Weak Base
pH of a Weak Base pH of .2 M of NH3 (weak base).
Lec 48 - Conjugate Acids and Bases
Conjugate Acids and Bases Introduction to conjugate acids and bases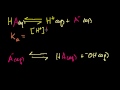
Lec 49 - pKa and pKb Relationship
pKa and pKb Relationship The pKa and pKb relationship between conjugate acids and bases (both of which are weak).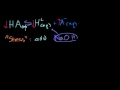
Lec 50 - Buffers and Hendersen-Hasselbalch
Buffers and Hendersen-Hasselbalch Buffers and the Hendersen-Hasselbalch equation
Lec 51 - Strong Acid Titration
Strong Acid Titration Strong acid titration and equivalence point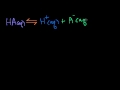
Lec 52 - Weak Acid Titration
Weak Acid Titration Equivalence point when titrating a weak acid
Lec 53 - Half Equivalence Point
Half Equivalence Point Figuring out the pKa of an unknown weak acid from the half equivalence point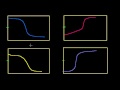
Lec 54 - Titration Roundup
Titration Roundup Making sure you fully understand titration curves
Lec 55 - Introduction to Oxidation States
Introduction to Oxidation States Oxidation and reduction. Oxidation states.
Lec 56 - More on Oxidation States
More on Oxidation States More practice calculating oxidation states
Lec 57 - Hydrogen Peroxide Correction
Hydrogen Peroxide Correction orrecting an error in the last video regarding hydrogen peroxide.
Lec 58 - Redox Reactions
Redox Reactions Oxidation reduction (or redox) reactions
Lec 59 - Galvanic Cells
Galvanic Cells Redox reactions to drive Galvanic Cells.
Lec 60 - Types of Decay
Types of Decay Alpha, Beta, Gamma Decay and Positron Emission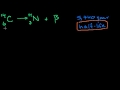
Lec 61 - Half-Life
Half-Life Introduction to half-life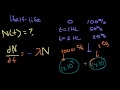
Lec 62- Exponential Decay Formula Proof ...
Exponential Decay Formula Proof (can skip, involves Calculus) Showing that N(t)=Ne^(-kt) describes the amount of a radioactive substance we have at time T. For students with background in Calculus. Not necessary for intro chemistry class.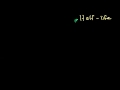
Lec 63 - Introduction to Exponential Decay
Introduction to Exponential Decay Introduction to Exponential Decay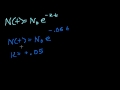
Lec 64 - More Exponential Decay Examples
More Exponential Decay Examples A few more examples of exponential decay.
Lec 65 - Macrostates and Microstates
Macrostates and Microstates The difference between macrostates and microstates. Thermodynamic equilibrium.
Lec 66 - Quasistatic and Reversible Proc ...
Quasistatic and Reversible Processes Using theoretically quasi-static and/or reversible processes to stay pretty much at equilibrium.
Lec 67 - First Law of Thermodynamics/ In ...
First Law of Thermodynamics/ Internal Energy First law of thermodynamic and Internal Energy
Lec 68 - More on Internal Energy
More on Internal Energy Getting more intuition of internal energy, heat, and work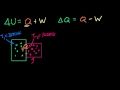
Lec 69 - Work from Expansion
Work from Expansion How a system can do work by expanding
Lec 70 - PV-diagrams and Expansion Work
PV-diagrams and Expansion Work Why work from expansion is the area under the curve of a PV-diagram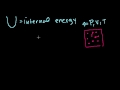
Lec 71 - Proof: U=(3/2)PV or U=(3/2)nRT
Proof: U=(3/2)PV or U=(3/2)nRT Conceptual proof that the internal energy of an ideal gas system is 3/2 PV.
Lec 72 - Work Done by Isothermic Process
Work Done by Isothermic Process Isothermic and Adiabatic processes. Calculating the work done by an isothermic process. Seeing that it is the same as the heat added.
Lec 73 - Carnot Cycle and Carnot Engine
Carnot Cycle and Carnot Engine Introduction to the Carnot Cycle and Carnot Heat Engine
Lec 74 - Proof: Volume Ratios in a Carno ...
Proof: Volume Ratios in a Carnot Cycle Proof of the volume ratios in a Carnot Cycle
Lec 75 - Proof: S (or Entropy) is a vali ...
Proof: S (or Entropy) is a valid state variable Prroof that S (or entropy) is a valid state variable.
Lec 76 - Thermodynamic Entropy Definitio ...
Thermodynamic Entropy Definition Clarification Clarifying that the thermodynamic definition of Entropy requires a reversible system.
Lec 77 - Reconciling Thermodynamic and S ...
Reconciling Thermodynamic and State Definitions of Entropy Long video explaining why entropy is a measure of the number of states a system can take on (mathy, but mind-blowing).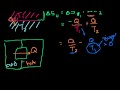
Lec 78 - Entropy Intuition
Entropy Intuition A discussion of what entropy is and what it isn't.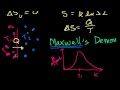
Lec 79 - Maxwell's Demon
Maxwell's Demon Maxwell's Demon: A thought experiment that seems to defy the 2nd Law of Thermodynamics
Lec 80 - More on Entropy
More on Entropy More clarification as to what entropy is and what entropy is not.
Lec 81 - Efficiency of a Carnot Engine
Efficiency of a Carnot Engine Definition of efficiency for a heat engine. Efficiency of a Carnot Engine.
Lec 82 - Carnot Efficiency 2: Reversing ...
Carnot Efficiency 2: Reversing the Cycle Seeing how we can scale and or reverse a Carnot Engine (to make a refrigerator)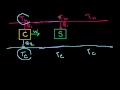
Lec 83 - Carnot Efficiency 3: Proving t ...
Carnot Efficiency 3: Proving that it is the most efficient Proving that a Carnot Engine is the most efficient engine
Lec 84 - Enthalpy
Enthalpy Understanding why enthalpy can be viewed as "heat content" in a constant pressure system.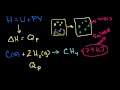
Lec 85 - Heat of Formation
Heat of Formation Standard heat of formation or standard enthalpy change of formation.
Lec 86 - Hess's Law and Reaction Enthalp ...
Hess's Law and Reaction Enthalpy Change Using Hess's Law and standard heats of formation to determine the enthalpy change for reactions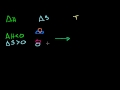
Lec 87 - Gibbs Free Energy and Spontaneity
Gibbs Free Energy and Spontaneity Intuition behind why spontaneity is driven by enthalpy, entropy and temperature. Introduction to Gibbs free energy.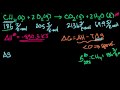
Lec 88 - Gibbs Free Energy Example
Gibbs Free Energy Example Determining if a reaction is spontaneous by calculating the change in Gibbs Free Energy.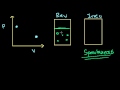
Lec 89 - More rigorous Gibbs Free Energy ...
More rigorous Gibbs Free Energy/ Spontaneity Relationship More formal understanding of why a negative change in Gibbs Free Energy implies a spontaneous, irreversible reaction.
Lec 90 - A look at a seductive but wrong ...
A look at a seductive but wrong Gibbs/Spontaneity Proof A look at why the "proof" of the relation between changes in Gibbs Free Energy and Spontaneity is wrong in many textbooks.
Lec 91 - Stoichiometry Example Problem 1
Stoichiometry Example Problem 1 Figuring grams of reactants and product produced from reaction of phosphorous and chlorine
Lec 92 - Stoichiometry Example Problem 2
Stoichiometry Example Problem 2 Stoichiometry Example Problem 2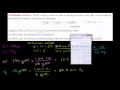
Lec 93 - Limiting Reactant Example Problem 1
Limiting Reactant Example Problem 1 Limiting Reactant Example Problem 1
Lec 94 - Empirical and Molecular Formula ...
Empirical and Molecular Formulas from Stoichiometry Empirical and Molecular Formulas from Stoichiometry
Lec 95 - Example of Finding Reactant Emp ...
Example of Finding Reactant Empirical Formula Example of Finding Reactant Empirical Formula
Lec 96 - Stoichiometry of a Reaction in ...
Stoichiometry of a Reaction in Solution Stoichiometry of a Reaction in Solution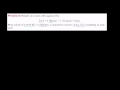
Lec 97 - Another Stoichiometry Example i ...
Another Stoichiometry Example in a Solution Another Stoichiometry Example in a Solution
Lec 98 - Molecular and Empirical Forumla ...
Molecular and Empirical Forumlas from Percent Composition Molecular and Empirical Forumlas from Percent Composition. Example 2.9 from Kotz Chemistry book
Lec 99 - Acid Base Titration
Acid Base Titration Using acid-base titration to find mass of oxalic acid
Lec 100 - Spectrophotometry Introduction
Spectrophotometry Introduction Spectrophotometry, Transmittance, Absorbance and the Beer-Lambert Law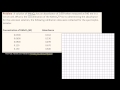
Lec 101 - Spectrophotometry Example
Spectrophotometry Example Spectrophotometry Example - Determining concentration based on absorbance
Lec 102 - Hess's Law Example
Hess's Law Example Hess's Law Example
Lec 103 - Vapor Pressure Example
Vapor Pressure Example Vapor pressure example using the Ideal Gas Law
Lec 104 - Change of State Example
Change of State Example Specific Heat Capacity and Enthalpy of Vaporization example
Chemistry
Source of these courses is khan
Videos on chemistry (roughly covering a first-year high school or college course).
khan Website: http://www.dnatube.com/school/khan
