Maintenance of pH: Buffer
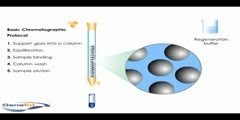
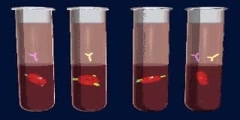
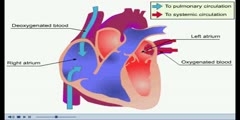
Buffer solutions are solutions which resist change in hydronium ion and the hydroxide ion concentration (and consequently pH) upon addition of small amounts of acid or base, or upon dilution. Buffer solutions consist of a weak acid and its conjugate base.
Channels: Biochemistry Chemistry (General)
Uploaded by: sana ( Send Message ) on 15-05-2007.
Duration: 0m 46s