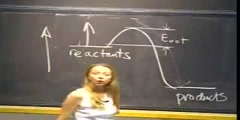Chemical Science - Chemical Equilibrium - Lecture 19
Principles of Chemical Science/n * Email this page/nVideo Lectures - Lecture 19/nTopics covered: /nChemical Equilibrium/nInstructor: /nProf. Catherine Drennan/nTranscript - Lecture 19/nIn this course, because I am a biological chemist now, I am going to give you some examples between chemistry and biology./nWe are going to teach you these principles of chemistry./nI am going to explain to you why that is relevant and how it applies to the more life sciences or biological areas to try to give you a sense of how I made this transition and being someone who did not like chemistry in high school to somehow who has now been a chemist for the rest of my life./nAnd so this is another important point that I like to make which is never say you really hate a subject, because I said I really do not like chemistry and became a chemist. I am doing chemistry for the rest of my life./nAnd that can happen to you, too./nSo be very careful what you say because you may discover later that that is really what you want to be doing. All right. What are we going to do in this second half of the course? We are going to first start out by finishing chemical equilibrium./nProfessor Ceyer started this. She led you from thermodynamics into chemical equilibrium and introduced you to the chemical equilibrium constant K. And so we are going to take it from there. And we will do a little review of what she covered on Monday as well./nThen we move into acid base equilibrium./nThis is a form of equilibrium. And from there to oxidation reduction reactions. And just from this connection between chemistry and biology, enzymes, which are the proteins in your body that catalyze all the reactions which allow you to walk around and breathe and all those kind of good things, most of these enzymatic reactions involve either an acid based kind of catalyzed reaction or oxidation reaction or both./nSo these are fundamental parts of how living systems work./nThen we cover transition metals, and I am going to explain during that part about metals in biology. A lot of enzymes require transition metals for their activity, so we will talk a little bit about that./nAnd then we end with kinetics in terms of the connection with biology, kinetics rates of reaction. What enzymes do is they work as catalysts to help speed up those reactions./nSo all of these topics are fundamental chemistry topics./nThey also apply to biology. The second half of the course is quite a bit different, really, from the first half of the course. In the first half of the course, you really get a lot of the basic principles, atomic theory, orbitals, bonding, Lewis structures, a lot of really fundamental things which in thermodynamics is what you need to know to be able to do the second half./nThe second half is really more applications of these basic principles./nAnd when you put this together, you get the fundamentals of chemistry that you need to go and study biochemistry, organic chemistry, inorganic biology, life sciences. These are the fundamental principles of chemistry which you get through in this half semester course./nSo it is somewhat of a different transition, and I just like to let people know ahead of time that it is going to feel like a little bit of a different course./nYou have a different instructor, and it is really somewhat different material./nIt builds on what you learned before, but it is really more applications. It is going to have a little bit of a different feel to it, so don't be surprised by that. Are there any questions? I will say the technical aspect of the course is the same./nYou have lecture notes, the way the problem sets are structured, everything like that is going to be the same. Use of calculators, all of that, the rules of the course do not change./nBut it is a little bit different material./nAre there any specific questions? And feel free to raise your hand. Maybe the TAs can help me if I don't always see that someone has a question, and phone extension, but usually email works best. We are doing a fairly smooth transition from the material on Monday to today./nWe're still on the topic in Chapter 9 in the book on chemical equilibrium./nAnd I just want to point out some things about the handouts that you've received. At the top of the handout, I have the chapter and page numbers available, so if you want to refer where this material is in your book that is always going to be listed at the top of the handout./nI also list on each handout at the top, the topics that are covered in that handout or in that lecture./nThis is just useful when you are going back and, say, reviewing for the final to see, oh, yes, on this day we covered these topics./nSo that is going to be listed at the top of each of these. And, as Professor Ceyer told you in the beginning of the course, these are lecture summaries that help you so you don't have to sort of scribble and write all the time that you can sort of sit back and listen more and write a few things./nBut, like Professor Ceyer, there are some blanks. Sometimes I am going to be wanting to ask you questions./nAnd so there are some blanks that you can fill in. So you should have a pen or pencil available because there are going to be places you are going to be wanting to write./nBut we provide these summaries to make it easier for you to not have to scribble constantly, especially with PowerPoint, so that you can really listen to what is going on. That is pretty much the same as what you've experienced./nBut if, of course, don't like those lecture summaries then put them away and you can take notes if that's how you feel that you learn better./nAll right. Chemical equilibrium. You were introduced to this a little bit on Monday./nYou moved from delta G into thinking about the relationship between delta G and K, the equilibrium constant. Basically, the point to remember about equilibrium is that it is a dynamic process. It's not that the reaction has stopped when it reaches equilibrium./nThe reactions are still going, but the rate of the forward reaction and the rate of the reverse reaction are equal so there is no net change in the composition of the material./nReactions are still going but there is no net change. Let's take a look at an example of what that means./nIf we take the reaction N2 gas plus H2 gas going to NH3 gas, a reaction with a delta G knot minus 32.90 kilojoules per mole, and if we consider what happens when the reaction starts, so we look at the concentrations of the reactants and products over time, at the beginning of the reaction you are only going to have your reactants./nYou would have hydrogen which would start and it would react so the concentration will change, it will decrease and then it will level off./nThe same will happen for nitrogen, the other reactant. It will start at a higher level, and then the concentration will drop and will level off./nAnd there will be no product in the beginning, but the product concentration will come up and then it will level off./nI want to make the point from the lecture notes. I cannot tell you how long trying to make the curves in PowerPoint. If anyone is really good at drawing sort of curve lines in PowerPoint you can see me later./nSo, if it looks sometimes like it is not really leveling off, that is a PowerPoint artistic issue./nAnd I will just say in general, for this course, that in terms of your ability to draw things on the exam, the standard for my parts of the exam, exam three, and my part of the final is that you have to draw as well as I draw./nThis is an extremely low bar in terms of drawing. And you will notice that what I went to college to study art was not on the list./nSo, I apologize for some drawing inabilities that may occur during this course./nBut the point here is that with the reactions that are starting at higher concentrations they are being consumed as they make product, product is coming up, but then all the lines flatten out. And what that means is that equilibrium has been reached./nThe reaction is still going on. It's going in the forward reaction, in the backward reaction, so there are arrows going forward and backward./nBut there is no net change so the concentrations are leveling off./nThe rate of the forward reaction is equal to the rate of the reverse reaction. We can think about this in a different way of what is going on here in terms of free energy./nIf we think about plotting free energy versus the progress of the reaction --/nSo free energy, or our friend delta G, versus the progress of the reaction./nIn the beginning you would have pure reactants./nAnd, at this point, delta G is going to be less than zero. It will be negative. So you will have a reaction that is spontaneous in the forward direction./nHere you would start up and it will come down./nYou will have a delta G less than zero until you reach some point where delta G equals zero./nSo you start here with pure reactants, and those pure reactants then will react and form some product. If you, however, start with pure products and you have no reactants, delta G will be greater than zero, so you will be spontaneous in the reverse direction of the reaction./nSo you will use up your products to form reactants./nAnd so here you go this way and here you are going to go down this way. And here you have a delta G that is greater than zero. So you are getting rid of your products. You started with pure products and you're making reactants until you get to this midpoint here where delta G equals zero./nAnd when delta G equal zero then you are at equilibrium./nThere is a relationship between delta Gs and the directions of these reactions. They are all moving toward equilibrium./nAlong with this then, we can think about the fact that delta G is going to be changing as the reactants and products change, as the composition of the reaction mixture changes./nWe start with pure reactants. You are going to have this negative delta G. At some points at equilibrium it will be zero. It is also possible to have a positive delta G if you start with pure products./nSo delta G is going to depend on your composition of your material./nAnd here is just a quick review from Monday. Professor Ceyer introduced you to this equation. The relationship between delta G, delta G knot and Q, which is the reaction quotient./nAgain, delta G is at any particular point in your reaction. Delta G knot has to do with the reactants and products in their standard states./nQ is the reaction quotient. And then R is the gas constant./nAnd T is temperature. She also introduced you to this on Monday, but I just want to review this. I think she actually just talked about the gaseous state, but the same equations and principles apply if you are talking about reactants in solution./nQ, this reaction quotient, the longer sort of definition of Q depends on the partial pressures of the reactants and the products or the concentrations if you are in solution./nShe showed you this equation of just A plus B going to C and D and said that the top of the reaction quotient is products and the bottom is reactants./nYou would have the partial pressure of C over some reference raised to the power of C, because you consider the stoichiometry of the reaction, and then it would be times D, the partial pressure of D over this reference raised to the power of D, again stoichiometry./nAnd then the same for the reactants. And over here, for solution, you're just talking about things in concentrations rather than in partial pressures./nBut then since the reference here would be one bar for partial pressure or one molar for concentration, you often see these with this reference dropped out of the equation, and you see Q as being equal to the partial pressure of C raised to the C, partial pressure of D raised to the D over partial pressure of A to the A and B to the B, or the same form in terms of concentrations./nThis is how you will be, in general, seeing it. And remember what your references are there. Again, she went over this on Monday./nWe will just look at it again. Also on Monday, she looked at this equation here and considered delta G when it is equal to zero, which means you are at equilibrium./nAnd, at that point, when delta G equals zero, Q, that reaction quotient equals K, which is the equilibrium constant. Q equals K, it has a new name when you are at equilibrium./nWe can take this equation then and substitute in zero, because at equilibrium delta G is zero./nAnd then this equation can be rewritten. You just bring the RT natural log of K to the other side. Again, K then is the equilibrium constant. It has the same form as Q./nBut you are only considering the concentrations at equilibrium./nIn the gaseous state, this looks just like Q, but these concentrations or partial pressures are the ones at equilibrium because K is the equilibrium constant. And, likewise, in solution, here you are just talking about the ratio of the products to the reactants at equilibrium./nI have these little equilibrium things to remind you that these are the concentrations at equilibrium./nShe also showed you, on Monday, that you can rearrange this equation. You can substitute in for delta G knot the minus RT natural log of K, because those are equal to each other./nAnd then you can rewrite that equation as delta G equals RT natural log of Q over K./nIf you're interested in this, this is a very useful equation. If you think about at what concentration are the reactants now, how does that compare to the concentrations at equilibrium? If you know where you are now, if you know Q now, you know what K is, you can think about the direction of the reaction./nYou can think about delta G. Is it negative or positive? Are you going the forward direction or are in you going in the reverse direction?/nThis is a very useful equation to predict the direction of the reaction depending on the composition at that moment compared to the composition at equilibrium./nIf Q is less than K, that is going to mean you will get a negative delta G, and you will be spontaneous in the forward direction./nBased on this equation, if Q is greater than K then delta G is going to be positive and you are going to go in the reverse direction./nAgain, this is very useful thinking about the relationship of Q and K and how that affects delta G. Let's look at an example then. We have our same reaction. Now we are given a value for K at 400 degrees C./nAnd we are given some partial pressures of the reactants and products./nAsk now are we at equilibrium here? If not, which direction is the reaction going to go? Let's figure out what Q is because we want to know the relationship between Q and K. That is going to tell us where we are./nAre we at equilibrium? What direction will the reaction go?/nAll right. Q. What is going to be on the top of the expression for Q? What goes on the top? Products or reactants? Products. And so we have one product here, so we are going to put our partial pressure of our product./nAnd then we have to consider the stoichiometry which would give us a two./nOn the bottom we have our reactants, so we will consider the partial pressure of N2 and the partial pressure of H2 cubed, again, the stoichiometry of the reaction./nThat is going to equal 1.1 squared over 5.5 times 2.2 cubed./nAnd if you work out the math there you get 2.1 times 10 to the minus 2, so that is Q. And K was listed as 1.9 times 10 to the minus 4 at the same temperature./nHere Q is greater than K. And so what direction will the reaction go?/nIt will go, as people said, toward reactants, or you could say in the reversed direction. So, NH3 will dissociate until equilibrium is reached./nAnd just one point about doing this, I have noticed people have come to you and said, oh, I knew the answer on the exam./nWhen I said left, what I meant was right. And since I have done that numerous times, I suggest that you say toward reactants or toward products or draw an arrow./nBecause usually then you actually write down what you mean./nMy personal tip is stay away from left and right on exams just in case, unless you are particularly good at that. But I have seen people be heartbroken when they knew the answer and just wrote the wrong direction./nHere we have an example where we were able to predict what direction the reaction would go by looking at Q and K./nLet's think about what else we can learn from K. The size of the equilibrium constant, the largeness of that value tells you something about the reaction./nWhat does K tell us?/nK, again, is equal to the products over the reactants at equilibrium. The size of K, whether it's greater or less than one, is going to tell us about the ratio of products to reactants at equilibrium./nWhen K is large, or we can say really what we're talking about is a K that is greater than one, you are going to have more products --/n-- than reactants at equilibrium, or high products./nThe products are going to be greater. If K is a big number, it has got to mean that there are more products than reactants at equilibrium, so you already know something when you know the value of K./nWhen K is small, when you have a K that is less than one then you are going to have low products or more reactants at equilibrium./nLet's look at an example for this, a case when K is greater than one./nLet's consider K greater than one, and let's look at a reaction for which that is true. Here is one. We have 2NO2 goes to N2O4./nK is 6.84 at 298 Kelvin. You have a delta G knot of minus 4.76 kilojoules per mole./nLet's look at a case where we start with just reactants. We have one bar of the NO2 gas and have no products. Let's consider then what is going to happen and calculate how this goes to equilibrium./nFor this, in this case, we actually have the K is 6.84 and we have the delta G knot minus 4.76 kilojoules per mole./nAgain, if we look at concentration, in this case we are talking about gases so we have partial pressures versus time, we are starting with just reactant, one bar of reactant./nAnd so the reactant will start up high and go lower as it reacts, our reactants./nThere will be no products when we start, but it will be created. And, eventually, when you reach equilibrium it will level off./nIn the beginning, with no products, Q is less than K./nBecause there are more products at equilibrium than there are now, which is there are none now, there are definitely going to be more at equilibrium./nWe have a negative delta G, and we will go in the forward direction of the reaction toward products, which is shown here. Reactants decrease, products form until you reach equilibrium. Now let's calculate what the concentration of these are going to be at equilibrium./nLet's calculate the equilibrium concentrations./nAnd our reaction, again, 2NO2 going to N2O4. Initially, we are going to have 1.000 bar and no product at all./nAs the reaction goes there will be a change, you will be making product, so you will be making product plus x, some amount of product will be formed./nAnd how much reactant will be lost during this change? Minus 2x./nAnd the 2 comes from the stoichiometry of the reaction. Then, at equilibrium, we would have 1.000 minus 2x and x. Now we can calculate K from this to find out what the concentrations of these gases are at equilibrium./nOr, find the concentrations from K, I should say./nK was 6.84 at this particular temperature. And so K is going to be equal to the products over the reactants. The partial pressure of N2O4 over the partial pressure of NO2 squared./nAgain, the stoichiometry of the reaction./nAnd then if we plug in these numbers, or plug in the equation x over 1.000 minus 2x squared./nAnd now you can take this and solve for x. Here, if you solve for x, it comes out at 0.381 bar. And so this is going to be your amount of product./nThat is the amount of N2, the partial pressure for the N2O4./nAnd we can go back and write that up here./nAnd then we can solve for the other one, 1.000 minus 0.381, sorry, 2 times 0.381, and that is going to give you 0.238 bar./nAnd that is going to be your partial pressure of your reactant at equilibrium, 0.238 bar. You see that when you have a K that is greater than one, you have more product than reactant at equilibrium./nAnd you can figure out that exact amount if you know how much you started with and you know K./nAnother thing to point out here is that in this case there is a relationship where you had a delta G knot that was negative 4.76 and that also there was this relationship between K and delta G knot./nIf you rearranged this equation to solve for K, you can see that if K is large then delta G knot is going to be negative and large./nIf you have a larger K, you are going to have a larger negative delta G. There is this relationship between delta G knot and K, and that can be derived from this equation./nYou can also go back and forth between these as well./nWhat that is going to translate into for a K larger than one, more products than reactants at equilibrium./nA couple more things that you should know about equilibrium expressions to be able to work all the problems./nAnd that is that if you want to add up two reactions and form a third reaction, you can also use the information about the equilibrium constants for each individual reaction to get an equilibrium constant for the summed reaction./nIf you add things together you multiply then the equilibrium constants./nCan I add these equations as such and get the third reaction on the bottom? What do I have to do? I have to multiply the second reaction by two./nI need to multiply that reaction by two. And then K3 is going to equal what in terms of K1 and K2?/nK1 times? Yeah, K1 times K2 times K2 or K1 times K2 squared. And then you can get K3 if you know those individual ones./nAnd let me just prove that that is true to you. We will give a little more practice writing out Ks and Qs./nK1 times K2 squared is going to give you K3. I am going to do products over reactants./nThe product is the partial pressure, PCL3 squared, and the reactant for the first one is going to be the partial pressure of P squared times the partial pressure of CL2 cubed./nAnd then K2 squared will have the partial pressure of PCL5 squared over the partial pressure of PCL3 squared, partial pressure of PCL2 squared./nAnd then, just as we did to get these equations to add up, you needed to cancel this one and cancel this one, those will cancel out here as well, and you will be left with an expression --/n-- like this./nAnd if we go back and double check that that is going to be correct then if we have --/nWait. This one is going to be Cl5, sorry./nActually, that is not canceling out appropriately./nIt is right./nOK, good. I am just having a problem canceling everything out. You should be able to add those together. All right. In the next couple of minutes we are going to talk about what happens if we take this system and then we apply stress to it./nWe have our conditions, we reach equilibrium and then a stress is applied. This is the principle of Le Châteier. And this is that a system in equilibrium that is subjected to a stress tends to react to minimize the stress./nAnd I find this a difficult concept for people who go to school at MIT and work at MIT./nAnd the reason why is that in this environment, when there is heavy stress applied, often the behavior is such to add more stress rather than minimize the stress./nYou have to think about this in a way as sort of other systems at other universities maybe down the street might work where a stress is applied and the reaction is, oh, let's minimize the stress. Let's make less stress./nThat is what Le Châteier's principle is about here./nIt turns out to be really useful because chemical reactions, unlike people at MIT, do tend to react in predictable ways to stress. And the chemical reactions react in such a way as to minimize the stress./nAnd this works almost all the time. You can predict the direction of the reaction based on the stress applied if you think about minimizing that stress./nLet's look at some examples. We can look at the simplest case which is adding or removing reactants or products./nWe will look at that, too. We looked at this reaction before. And when you talk about chemical equilibrium, this is the reaction you study, and we will be thinking about this on Friday as well./nHere, again, you start with hydrogen and nitrogen and you're making ammonia./nYou start with just the reactants, no product, and they react and then they level off when equilibrium is reached. What happens then if we're at equilibrium, someone comes along and adds more H2? Which direction will the reaction go? What do you guess? You want to minimize the stress./nMinimize the amount of H2./nIt will go to our products. You will use up more N2 and you will make more product. And they will react and then will come to equilibrium again. And the net composition will have the same ratio once you reach equilibrium./nWhat if you added more product? What is going to happen? It will go in the reverse direction./nAnd so you will end up making more reactants, both H2 and N2, until you reach a new equilibrium again./nYou have the same ratios as you did before. We can think about this. You tend to minimize the stress of the reaction. You add more reactant, you use it up and make more product. You add more product, you use it up and make more reactants./nThe reaction will shift toward product, in this case, when we are adding more hydrogen./nAnd we can think about this in terms of Q and K again. If we think about this and we add more reactant then momentarily Q will drop below K. And so the reaction, when that happens, you will have a negative delta G./nRecall this is our predictive equation that we saw earlier. We can think about the ratio of Q to K in terms of the sign of delta G./nSo with Q less than K, negative delta G, we make more product. This makes sense./nYou came up with the right answer without thinking about this equation, I think, but we can also rationalize it from this equation. And then we saw this before, adding more reactant, making more product, shifting it toward products to the right./nWe also considered adding more product./nAgain, you can rationalize in terms of Q and K and delta G. As you add more product, Q is momentarily larger than K, there is more products over reactants, Q is bigger, so that is going to mean a positive delta G which means that it is spontaneous in the reverse direction so you make more reactants./nYou can think about this in terms of the equation, or you can think about it in terms of minimizing stress./nAgain, we saw this example. We added more product. We made more reactants. To compensate, you add more product, it removes the product./nFinally, what would happen in this case if we remove product? What happens? Right, you would make more product. Again, you can think about this in terms of Le Châteier, you remove product./nTo compensate for that stress you make more product./nOr, you can think about it in terms of Q and K. If you remove product then you are going to have a Q that is less than K because you just pulled product out, so now you have less product than you did at equilibrium./nAnd with Q less than K, you have a negative delta G. And so your system will shift with a negative delta G to the right. It will be spontaneous in the forward direction./nSo, Le Châteier's principle or this equation are really good at predicting stress./nAnd we will continue on Friday talking about Le Châteier and move on to think about other forms of shifting the reaction based on stresses that you add to the system.
Channels: Chemistry (General)
Tags: Chemical Equilibrium
Uploaded by: mitlectures ( Send Message ) on 16-04-2009.
Duration: 42m 55s